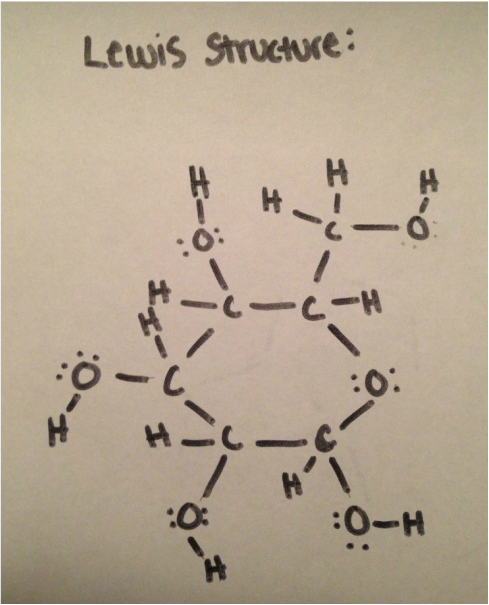
Lewis structure
The Lewis Structure of D-Glucose consists of 6 carbon elements, 6 hydrogen elements, and 12 hydrogen elements. The molecular formula of D-Glucose is C6H12O6. The Lewis Structure consists of single bonds between the elements, meaning that the when the valence electron level of one element isn't full, it pairs up with another element that either needs to loose or gain an electron to make the atom more stable.
nature of bonds involved
The bonds of D-Glucose consist of:
C-H |2.55-2.1|=.45 very covalent
O-H |3.44-2.1|=1.34 polar covalent
O-C |3.44-2.55|=.89 polar covalent
C-H |2.55-2.1|=.45 very covalent
O-H |3.44-2.1|=1.34 polar covalent
O-C |3.44-2.55|=.89 polar covalent
Vsepr form
The vsepr structure of C6H12O6 is cyclical.